Abstract
Background: Monoclonal B-cell lymphocytosis (MBL) is a benign precursor condition to chronic lymphocytic leukemia (CLL). Clonal hematopoiesis of indeterminate potential (CHIP) is an age-related condition characterized by accumulation of somatic mutations in the blood. Both conditions involve low level clonal populations in the blood. Review of the literature identified one prior abstract evaluating CHIP in patients with MBL. This study found CHIP in 17% of 251 patients with CLL (median age 63), 12% of 74 patients with MBL (median age 61.5), and 8% of 246 unaffected relatives (median age 54). CHIP was not more common in patients with MBL compared to CLL (Boddicker. Blood. 2018;132(S1): 3116). In this study, we focused on myeloid CHIP (M-CHIP) specifically, to distinguish CHIP mutations from MBL/CLL related mutations. Our objective was to characterize the prevalence of M-CHIP in patients with MBL compared to patients with untreated CLL.
Methods: We performed a retrospective study including 44 patients with MBL and 749 patients with untreated CLL enrolled in a prospective CLL biobank at Dana-Farber Cancer Institute who had a 95-gene targeted next-generation sequencing (NGS) panel performed (J Mol Diagn. 2016;18:507). We created a list of 56 M-CHIP candidate genes using published criteria on M-CHIP (Nat Med. 2021;27:1921) and excluded 8 overlap genes that were more common in CLL than M-CHIP (SF3B1, TP53, BRAF, KRAS, BCOR, NRAS, CREBBP, SETD2; Knisbacher. Nat Genetics. In press). We performed a sensitivity analysis in which we only excluded overlap genes among the top 5 driver genes in CLL (specifically, only SF3B1, TP53). We analyzed the association of M-CHIP in patients with MBL vs. untreated CLL patients using logistic regression controlling for IGHV mutational status. We also performed an exploratory landmark survival analysis from time of NGS panel to assess the relationship between CHIP and progression from MBL to CLL.
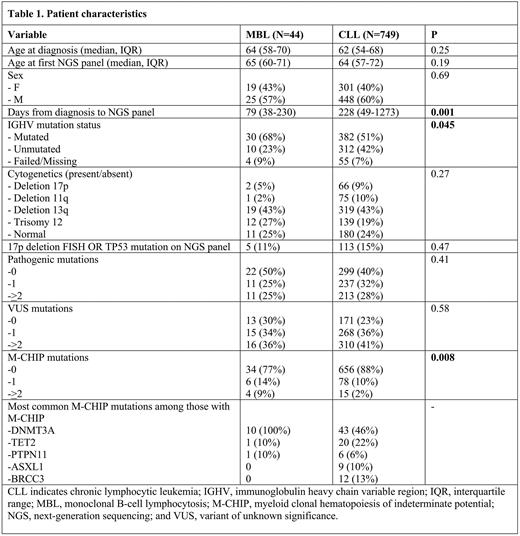
Results: In our cohort of 793 patients, there were no significant differences in age at diagnosis or NGS panel, sex, and cytogenetics, for patients with MBL vs. untreated CLL (Table 1). Median age at time of NGS panel for patients with MBL vs. untreated CLL was 65 (interquartile range (IQR) 60-71) and 64 (IQR 57-72) respectively. 43% vs. 40% of patients were female, respectively. Patients with MBL and untreated CLL patients were equally likely to have pathogenic mutations (50% vs. 60%, p=0.2) or VUS (70% vs. 77%, p=0.3) on the NGS panel, and had a similar total number of mutations (Table 1). Among patients with MBL, the most common pathogenic mutations were in DNMT3A (27%) and NOTCH1 (9%) and the most common VUS were in ATM (18%) and BCOR (14%). Among patients with untreated CLL, the most common pathogenic mutations were in NOTCH1 (15%) and SF3B1 (13%) and the most common VUS were in ATM (17%), CREBBP (6%), and TET2 (6%).
The prevalence of M-CHIP was significantly greater among patients with MBL compared to patients with untreated CLL (23% vs. 12%, OR 2.2, 95% CI 1.1-4.7, p=0.034). However, when we used the more sensitive definition of M-CHIP, it was no longer more common in patients with MBL than CLL (25% vs. 18%, OR 1.7, 95% CI 0.8-2.2, p=0.15). Notably, the median age of patients with M-CHIP with MBL vs. untreated CLL was similar (70 (IQR 65-74) vs. 67 (IQR 61-74) (p=0.53)). Among patients with M-CHIP, patients with MBL were more likely to have DNMT3A mutations compared to patients with CLL (100% vs. 46%, p=0.001).
In an exploratory analysis with a median follow-up time of 152 days (IQR 28-639), CHIP as a binary predictor was not associated with progression from MBL to CLL (HR 0.9, 95% CI 0.2-4.4, p=0.9). This analysis was limited in power due to a low number of progression events (N=9) and limited follow-up duration.
Conclusions: When using a strict definition for M-CHIP, we found that the prevalence of M-CHIP was significantly greater in patients with MBL (23%) than in patients with untreated CLL (12%). However, this association did not persist when using a more sensitive definition of M-CHIP allowing more overlap between CLL driver genes and M-CHIP. Patients with CLL are more likely to have CLL driver genes than patients with MBL, explaining this discrepancy. This illustrates the challenges in M-CHIP categorization for patients with MBL/CLL, and further research is ongoing in which blood from patients with MBL is sorted into myeloid and lymphoid compartments to allow more definite categorization.
Disclosures
Getz:Scorpion Therapeutics: Consultancy, Current equity holder in publicly-traded company, Other: Founder; IBM: Research Funding; Pharmacyclics: Research Funding; SignatureAnalyzer-GPU: Patents & Royalties; MSMuTect: Patents & Royalties; MSMutSig: Patents & Royalties; MSIDetect: Patents & Royalties; POLYSOLVER: Patents & Royalties. Wu:Pharmacyclics: Research Funding; BioNTech: Current equity holder in publicly-traded company. Davids:AbbVie: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Travel expenses, Research Funding; Adaptive Biotechnologies: Consultancy, Membership on an entity's Board of Directors or advisory committees; Ascentage Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; AstraZeneca: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; BeiGene: Consultancy, Membership on an entity's Board of Directors or advisory committees; Bristol-Myers Squibb: Consultancy, Membership on an entity's Board of Directors or advisory committees; Eli Lilly and Company: Consultancy, Membership on an entity's Board of Directors or advisory committees; Genentech: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees; Merck: Consultancy; Novartis: Research Funding; Ono Pharmaceuticals: Consultancy; Research to Practice: Honoraria; Takeda: Consultancy; TG Therapeutics: Consultancy, Research Funding; Verastem: Consultancy, Research Funding. Brown:Abbvie: Consultancy; Acerta/Astra-Zeneca: Consultancy; Beigene: Consultancy; Bristol-Myers Squibb/Juno/Celgene: Consultancy; Catapult: Consultancy; Genentech/Roche: Consultancy; Janssen: Consultancy; MEI Pharma: Consultancy; Morphosys AG: Consultancy; Novartis: Consultancy; Pfizer: Consultancy; Rigel: Consultancy; Gilead: Research Funding; Loxo/Lilly: Research Funding; Verastem/SecuraBio: Research Funding; Sun: Research Funding; TG Therapeutics: Research Funding; Invectys: Other: served on the data safety monitoring committee.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal